Publications
Roles of Mo Surface Dopants in Enhancing the ORR Performance of Octahedral PtNi Nanoparticles
Qingying Jia, Zipeng Zhao, Liang Cao, Jingkun Li, Shraboni Ghoshal, Veronica Davies, Eli Stavitski, Klaus Attenkofer, Zeyan Liu, Mufan Li, Xiangfeng Duan, Sanjeev Mukerjee, Tim Mueller, Yu Huang
Nano Lett. 18, 798-804 (2018)
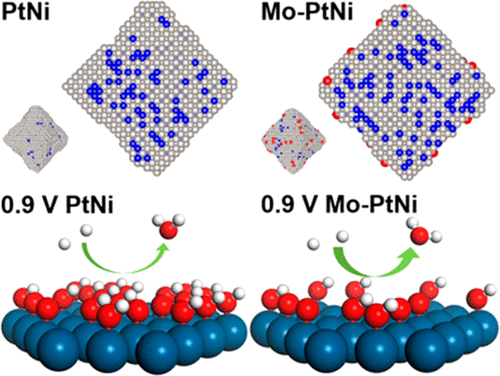
Doping with a transition metal was recently shown to greatly boost the activity and durability of PtNi/C octahedral nanoparticles (NPs) for the oxygen reduction reaction (ORR), but its specific roles remain unclear. By combining electrochemistry, ex situ and in situ spectroscopic techniques, density functional theory calculations, and a newly developed kinetic Monte Carlo model, we showed that Mo atoms are preferentially located on the vertex and edge sites of Mo–PtNi/C in the form of oxides, which are stable within the wide potential window of the electrochemical cycle. These surface Mo oxides stabilize adjacent Pt sites, hereby stabilizing the octahedral shape enriched with (111) facets, and lead to increased concentration of Ni in subsurface layers where they are protected against acid dissolution. Consequently, the favorable Pt3Ni(111) structure for the ORR is stabilized on the surface of PtNi/C NPs in acid against voltage cycling. Significantly, the unusual potential-dependent oxygen coverage trend on Mo-doped PtNi/C NPs as revealed by the surface-sensitive Δμ analysis suggests that the Mo dopants may also improve the ORR kinetics by modifying the coordination environments of Pt atoms on the surface. Our studies point out a possible way to stabilize the favorable shape and composition established on conceptual catalytic models in practical nanoscale catalysts.
UCLA, Department of Chemistry and Biochemistry
607 Charles E. Young Drive East, Box 951569
Los Angeles, CA 90095-1569
E-mail: xduan@chem.ucla.edu
607 Charles E. Young Drive East, Box 951569
Los Angeles, CA 90095-1569
E-mail: xduan@chem.ucla.edu







